Study Title: The effect of two pre-analytical variables (type of collection tube and time to analysis) on the detection and enumeration of Circulating Tumor Cells (CTCs)
This page: https://nciphub.org/groups/nci_physci/wiki/PSON0007
The data set: ftp://caftpd.nci.nih.gov/psondcc/CTC/
Study Contact: Peter Kuhn email: pkuhn@usc.edu
Overview: The goal is to characterize the effect of two pre-analytical variables, namely five blood collection tubes (BCT), Streck, EDTA, Heparin, Citrate ACD and CellSave, and four times-to-assay (TTA) on the bench, 24 and 72 hours, relevant to the enumeration of circulating tumor cells (CTCs) and specific high-content screening (HCS) measurements. Enumeration and characterization of HD-CTC will determine the best combination of BCT and TTA that will subsequently be compared to the CellSearch® System
High Definition-Single Cell Analysis assay (HD-SCA)
A high-content screening (HCS) methodology is being used to characterize multiple parameters at the single cell level in conjunction with a HCS triaging concept to identify events of interest. Developed at the Kuhn laboratory of the University of Southern California (formerly at The Scripps Research Institute), the HD-SCA assay is an inherently non-enrichment assay providing multiple overall opportunities as a fluid biopsy. Its expanded use requires a detailed understanding of pre-analytical variables related to biospecimen collection, handling and processing.
The approach used is essentially immuno-histochemical (IHC). We use a small molecule nuclear stain, a pan-cytokeratin Ab with a fluorescent dye and a CD-45 pre-conjugated AB (with a fluorescent dye). The staining is performed on cells that are adhered to glass slides which have been stored in a -80 C freezer. After thawing the slides, they are rinsed and placed into the auto-stainer where consistent amounts of reagent (and appropriate wash steps) are applied. After the staining process, the slides are cover-slipped using a glycerol-based mounting medium and the coverslip is sealed to the glass slide. The slides are then added to the scanning queue. Scanning is performed using our custom-built whole slide imagers controlled by in-house software. Auto-focus and auto-exposure values (in each of the 4 color channels) are calculated prior to start of a slide scan. Then each slide is scanned in 4 colors and the data is moved to an HPC computer cluster where the initial image analysis is performed resulting in a web-based report that displays the candidate cells. This report is then used by a trained technician to classify the candidate cells.
Recruitment Plan
A stand-alone trial was initiated: Optimizing Parameters and Techniques in Circulating Tumor Cell Collection (OPTICOLL). In this protocol, early stage patients are enrolled prior to the initiation of treatment, without constraints on the type of treatment that will be received. There are two blood draws collected per early patient, one prior to treatment and the other 3 weeks post treatment. For late-stage patients, there are no constraints on what prior therapies had been used. There is the potential for up to 12 draws per patient for late stage patients. A total of 90 breast cancer patients will be enrolled in the study from three clinical sites. Patients with early stage disease will have 2 study related blood draws, whereas up to 12 study related blood draws will be obtained for patients with metastatic disease over a 2-year period.
Experimental Design
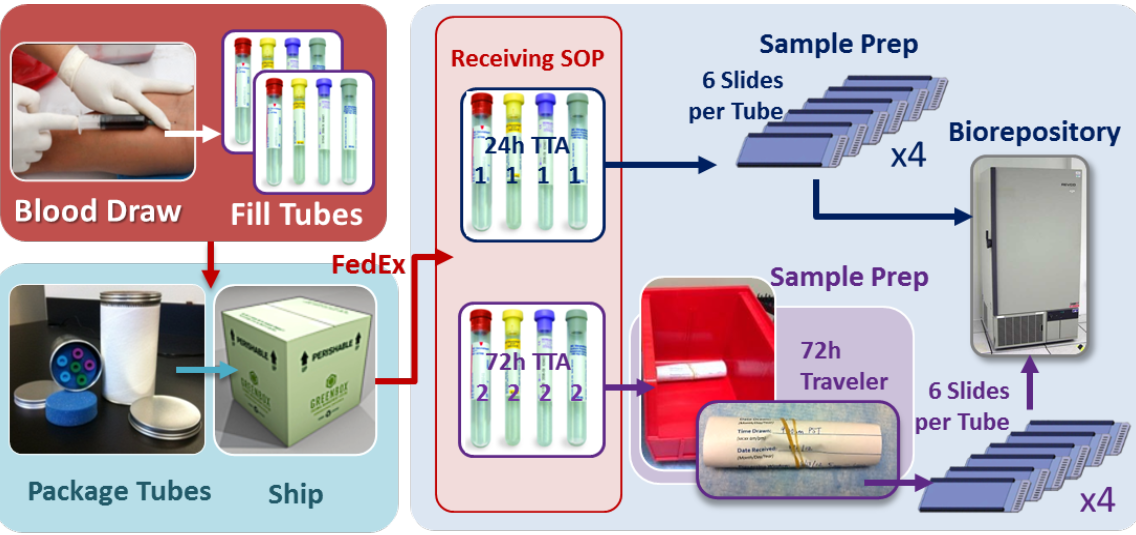
Blood from patients is drawn into a syringe and distributed amongst the different blood collection tube types. Variables such as anatomic draw location, needle gauge and tube fill number are tracked with a standardized questionnaire. All BCTs are then packaged and sent to the central laboratory. The 24h TTA tubes are processed and stored in the -80°C biorepository upon arrival. The 72h TTA tubes are stored in a designated location until 68-76 hours after the time of draw. Six slides are created from each BCT and all slides are stored in the biorepository until analysis.
Blood specimens from research subjects are sent by FedEx overnight to USC for sample preparation and storage into the -80°C biorepository. At the time of draw at the clinical site, information about the blood draw parameters is collected. For Veridex specimens, samples collected in CellSave™ tubes will be forwarded to a commercial laboratory for CellSearch® analysis. Experiments for copy number variation (CNV) will require cell picking at USC with the individual cells being shipped to Cold Spring Harbor Laboratory for analysis.
Whole-genome copy number variation (CNV) analysis in individual CTCs
Ten CTCs from blood draw C1_S025.1 were individually harvested into PCR tubes. After single-cell whole genome DNA amplification (WGA, Qiagen) and library construction, the genome was sequenced (Illumina) and analyzed for genomic copy number variations (CNV) as described in: Dago AE, Stepansky A, Carlsson A, Luttgen M, Kendall J, et al. (2014) Rapid Phenotypic and Genomic Change in Response to Therapeutic Pressure in Prostate Cancer Inferred by High Content Analysis of Single Circulating Tumor Cells. | PLOS ONE 9(8): e101777.
In short the informatics process consisted of: 1) Deconvoluting the sequence reads based on barcodes 2) Mapping the reads to the human genome (Hg19) and removing PCR duplicates 3) Normalizing for G-C content and estimating copy number using the CBS segmentation algorithm
In addition to the CTC concentrations and CTC images (10X magnification), the pre-analytical draw data, the HD-SCA high content data (relative nuclear size and relative cytokeratin intensity), and the CellSearch® results are provided.
RESULTS
The entire data set for this study includes: CTC concentrations, CTC images (10X magnification), the blood draw parameters, HD-SCA high content data for individual CTCs (relative nuclear size and relative cytokeratin intensity) and the CellSearch® comparison results. Also included is the copy number analysis of ten individual CTCs from a single blood draw. The copy number profile data as a function of chromosome genomic location, along with corresponding high-resolution fluorescence images (40X magnification) for each CTC are provided. This data package can be downloaded at ftp://caftpd.nci.nih.gov/psondcc/CTC/ (See the READMe.txt file for additional details on the submitted data package.)
CTC Enumeration Results Summaries
Experiment 1. Effect of Blood Collection Tube type on CTC enumeration using the HD-SCA assay.
The purpose of this experiment is to characterize the the effect of using different types of blood collection tubes (BCTs) on the the HD-SCA assay. The four tube types examined are Streck, EDTA, Heparin, and Citrate ACD. The time to assay for this experiment is 24 hours. The best performing tube in this experiment will be used in Experiment 2. Download spreadsheet: ftp://caftpd.nci.nih.gov/psondcc/CTC/ancillary/countsPerBCT.xls
Experiment 2. Effect of Time to Assay on CTC enumeration using the HD-SCA assay.
The purpose of this experiment is to characterize the effect of Time to Assay on the enumeration of CTCs using the HD-SCA assay. The Streck tube was used for this experiment. The times to Assay measured were 24 hours and 72 hours. The best performing time to assay will be used for Experiment 3. Download spreadsheet: ftp://caftpd.nci.nih.gov/psondcc/CTC/ancillary/countsPerTTA.xls
Experiment 3. Comparison of the HC-SCA assay and the Veridex CellSearch® Assay for enumeration of CTCs.
Concentration of CTCs enumerated comparing results of HD-SCA with those obtained using CellSearch® methodology. Download spreadsheet:ftp://caftpd.nci.nih.gov/psondcc/CTC/ancillary/countsPerAssayType.xls
Users who wish to browse the 10X fluorescent images of the identified CTCs can find thumbnail images in the ancillary directory: ftp://caftpd.nci.nih.gov/psondcc/CTC/
Data usage policy
The data contained within the PS-ON DCC is based on several research projects and is intended to be rapidly and constantly updated for the research community to access and use. The NCI requests that any data users:
- Inform the data submitters about the intention to submit a publication that uses PS-ON DCC data.
- Include the following statement in any publications resulting from the use of this specific PS-ON DCC data set: Data used in this publication were generated by projects sponsored by the NCI Center for Strategic Scientific Initiatives (CSSI).